How to create and sustain clinical-research partnerships
In The BMJ, NIH Collaboratory provides guide to success for pragmatic trials
SEATTLE—Pragmatic clinical trials—real-life tests done in real-world settings—are increasingly important for answering pressing questions about how best to deliver health care. But these pragmatic trials require close collaboration between two professional groups who often have contrasting styles. One group is researchers, who follow structure like classical musicians. The other is and health care providers and administrators, who may need to improvise like jazz musicians. How in the world can such disparate players make beautiful music together?
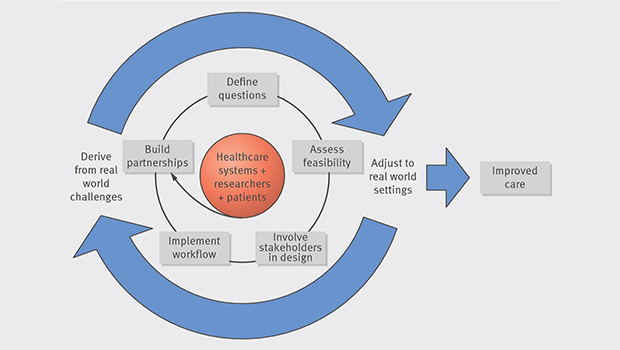
“The key is not just to waltz in—but instead to get in tune with each other, make compromises, and ‘rehearse’ with pilot periods,” said Karin E. Johnson, PhD, a research associate at Group Health Research Institute. She is the first author of “A guide to research partnerships in pragmatic clinical trials” in The BMJ, which stresses these steps: Build partnerships, define clinically important questions, assess feasibility, involve stakeholders in study design, and develop study workflows.
The guide is a product of interviews with pragmatic clinical trial experts, including people involved in demonstration pragmatic clinical trials supported by the Health Care Systems Research Collaboratory of the U.S. National Institutes of Health (NIH) and the REDUCE MRSA trial. The guide highlights the following lessons about building strong research partnerships between health researchers and health care systems:
- Participating in pragmatic clinical trials can provide health care systems with evidence and tools to improve health care—and researchers with chances to do high-impact studies.
- Pragmatic clinical trials answer questions that matter to health care systems. So clinicians, health care managers, health information technology, and clinical operations staff—and, increasingly, patients—should be involved in designing studies.
- Successful pragmatic clinical trials start with a strong researcher-health care system partnership. They go through rigorous, objective evaluation of the partner health care system(s)’s ability to participate. And they produce long-term scientific relationships—and evidence about sustainable ways to improve care.
Pragmatic clinical trials are comparative effectiveness studies conducted in real-world settings to answer questions that matter to patients, clinicians, and health care decision makers, Dr. Johnson explained. They include some randomized trials. By contrast, explanatory clinical trials study how treatments or interventions work in carefully controlled settings and study populations, often to investigate a biological hypothesis or test a drug or device to meet regulatory requirements.
“This guide is a product of the Collaboratory’s Health Care Systems Interactions Workgroup,” said Dr. Johnson’s coauthor Eric. B. Larson, MD, MPH, the executive director and a senior investigator at Group Health Research Institute and Group Health’s vice president for research. “We’re excited about sharing the valuable lessons that we’ve learned about how best to create and sustain research partnerships for pragmatic clinical trials in a widely read and prestigious journal like The BMJ.”
Members of the NIH’s Health Care Systems Research Collaboratory overlap with those of the HMO Research Network, which includes 18 research centers, each associated with a health care delivery system. When this guide was presented at the 2014 annual conference of the HMO Research Network, it was chosen as its first annual “paper of the year.”
A cooperative agreement (U54 AT007748) from the NIH Common Fund for the NIH Health Care Systems Research Collaboratory supported this work.
Drs. Johnson and Larson’s coauthors are Gregory E. Simon, MD, MPH, a Group Health psychiatrist and senior investigator at Group Health Research Institute; Chris Tachibana, PhD, scientific editor and writer, Julie Richards, MPH, and Ella Thompson, project managers, all at Group Health Research Institute; Gloria Coronado, PhD, a senior investigator at Kaiser Permanente Center for Health Research, in Portland, OR; Laura Dember, MD, a professor of medicine at the University of Pennsylvania Perelman School of Medicine in Philadelphia; Russell Glasgow, PhD, associate director of the Colorado Health Outcomes Program at the University of Colorado School of Medicine in Aurora; Susan Huang, MD, MPH, an associate professor and medical director of epidemiology and infection prevention at the University of California Irvine School of Medicine; Paul J. Martin, MD, medical director of Clinical Research Support at the Fred Hutchinson Cancer Research Center in Seattle; Gary Rosenthal, MD, a professor of internal medicine at the University of Iowa in Iowa City; Ed Septimus, MD, medical director, Infection Prevention and Epidemiology, Clinical Service Group, Hospital Corporation of America in Nashville; Leif Solberg, MD, associate medical director for care improvement research at HealthPartners Institute for Education and Research in Bloomington, MN; and Jerry Suls, PhD, a senior scientist at the National Cancer Institute in Bethesda, MD.
NIH’s Health Care Systems Research Collaboratory
Supported by the Common Fund at the National Institutes of Health (NIH), the Health Care Systems Research Collaboratory is intended to improve the way clinical trials are conducted by creating a new infrastructure for collaborative research. The ultimate goal is to ensure that healthcare providers and patients can make decisions based on the best available clinical evidence. The NIH HCS Research Collaboratory also supports the design and rapid execution of several high-impact Pragmatic Clinical Trial Demonstration Projects that will address questions of major public health importance that engage health care delivery systems in research partnership.
About Kaiser Permanente
Kaiser Permanente is committed to helping shape the future of health care. We are recognized as one of America’s leading health care providers and not-for-profit health plans. Founded in 1945, Kaiser Permanente has a mission to provide high-quality, affordable health care services and to improve the health of our members and the communities we serve. We currently serve more than 12.4 million members in eight states and the District of Columbia. Care for members and patients is focused on their total health and guided by their personal Permanente Medical Group physicians, specialists and team of caregivers. Our expert and caring medical teams are empowered and supported by industry-leading technology advances and tools for health promotion, disease prevention, state-of-the-art care delivery and world-class chronic disease management. Kaiser Permanente is dedicated to care innovations, clinical research, health education and the support of community health. For more information, go to: kp.org/share.
FOR IMMEDIATE RELEASE
Media contact
For more on Kaiser Permanente Washington Health Research Institute news, please contact:
Bianca DiJulio
bianca.s.dijulio@kp.org
206-660-8333
After-hours media line: 206-448-4056
Co-Researchers
,
Gregory E. Simon, MD, MPH
Senior Investigator
KPWHRI; Psychiatrist, Washington Permanente Medical Group


