Research versus COVID-19
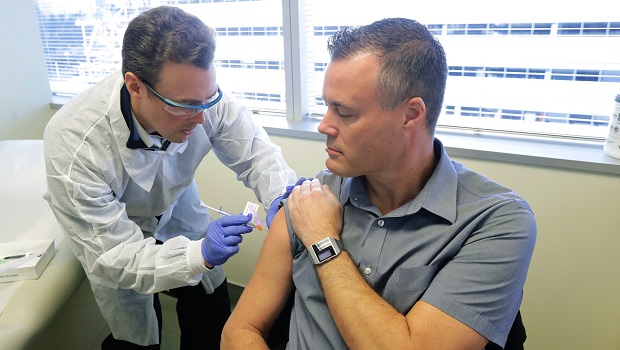
Pharmacist Michael Witte, left, gives Neal Browning a shot of an investigational vaccine for COVID-19 on March 16, the day that the phase 1 clinical trial launched at KPWHRI. (AP Photo/Ted S. Warren)
From clinical trials to technical support, Dr. Rita Mangione-Smith describes how Kaiser Permanente Washington researchers are fighting the novel coronavirus.
By Rita Mangione-Smith, MD, MPH, Kaiser Permanente Washington Health Research Institute executive director and Kaiser Permanente Washington vice president for research and health care innovation
With so much changing around us, it seems hard to believe that it was just 2 months ago that I got a call from Lisa Jackson, MD, MPH, who leads vaccine research here at Kaiser Permanente Washington Health Research Institute (KPWHRI). Her team had been selected to run the world’s first human trial of a vaccine against the novel coronavirus that had just emerged in China. Lisa needed the OK from Kaiser Permanente Washington to proceed with the study, sponsored by the National Institute of Allergies and Infectious Diseases (NIAID).
Approval from our health plan and medical leaders came fast. We learned that the vaccine does not contain any part of the virus, so it could not cause infection to those who would eventually volunteer for the trial nor to our staff who would do the work. We also understood that this work is crucial to the fight against COVID-19 worldwide.
What many of us may not have realized, however, was how quickly our own lives were about to change. By the time our vaccine research team gave the first injection on March 16, Kaiser Permanente Washington and other health care systems in our region were mobilizing for an unprecedented surge in illness among patients infected by the virus. Like many workers in our region, most of our Institute’s 300 employees were preparing to follow Governor Jay Inslee’s guidance—and eventual order—to work from home. Only those working in jobs essential to COVID-19-related clinical trials would keep coming to the institute.
We were following the advice of scientists from the Seattle Flu Study, including KPWHRI Associate Investigator and epidemiologist Mike Jackson, PhD, who had been sharing their findings with health leaders and the national media. Their evidence showed how the new coronavirus was spreading through our communities, making it clear that social distancing would be our best strategy for “flattening the curve” of disease transmission.
Collective action sparks hope
News of the virus’s rapid spread and enormous toll has been extremely sobering. And yet, amid this troubling news, our community’s determination to defeat the virus provides glimmers of hope. Our collective efforts around social distancing and work-from-home requirements appear to be slowing the virus’ spread and may soon result in reduced rates of COVID-19 hospitalizations and deaths in our state. But even if we show success, such improvements should not be a signal to change strategies; relaxing these measures too soon would likely bring about a resurgence of the illness. We must be willing to stay the course.
I also see signs of hope among those working in health care delivery at Kaiser Permanente and throughout the community. The dedication and courage of providers caring for Kaiser Permanente members and others throughout this crisis is truly inspiring. Our providers are not only meeting their commitment to provide high-quality, affordable health care, they are quickly adapting to a new “Virtual First” care model, which is helping to keep our patients and staff members safe. By the last week of March, 90% of all primary care visits and 75% of all specialty care visits in the Kaiser Permanente Washington system were done virtually.
New trial to launch soon
I am also inspired by the energetic response to the crisis from KPWHRI faculty and staff who immediately began applying the Institute’s research capabilities to the fight against COVID-19. In addition to our vaccine trial, we’ll soon be launching the Washington Respiratory Virus Study led by Mike Jackson and funded by the Centers for Disease Control and Prevention. Building on Mike’s years of experience studying influenza, this new trial will randomly reach out to 1,000 Kaiser Permanente Washington members who seek care for acute respiratory illness, sending them a self-administered nasal-swab test kit that they will return by mail. Those who test positive for COVID-19 will be immediately contacted with instructions for further care. Data collected in the study will help us to:
- better track the spread of COVID-19, flu, and other respiratory infections in our communities;
- examine the range of symptoms people are experiencing with various respiratory infections, including COVID-19;
- track how respiratory viruses evolve over time, so that we can do a better job predicting and preventing their spread; and
- learn how well certain drugs work to treat COVID-19.
Meanwhile, our faculty and staff are providing a variety resources and support to the Kaiser Permanente Washington care-delivery system. Examples include:
- Programming that used predictive analytics to identify Kaiser Permanente Washington Medicare-age members at highest risk for complications of COVID-19. Using this data, Kaiser Permanente Washington care providers are proactively reaching other to these members offering care and support.
- Consultation to Washington state’s COVID-19 Health System Response Management team in developing a database to track hospital and ICU utilization statewide.
- Production of videos needed for virtual patient education regarding diabetes self-care and self-testing for COVID-19.
- Working with KP Washington’s newly formed Business Command Center to understand, improve, and evaluate the “Virtual First” care model that is quickly evolving from the system’s rapid COVID-19 response.
- Rapid reviews of scientific evidence related to COVID-19 care.
More studies in pipeline
In addition, our MacColl Center for Health Care Innovation is working with Kaiser Permanente Washington Community Health and various community partnerships to identify and share resources with care providers in the region’s federally funded safety net community clinics. Examples include sharing resources such as in-clinic workflows and other clinical protocols for COVID-19, and leading collaboratives to plan for emerging social needs resulting from the pandemic.
Importantly, our scientific teams are now hard at work designing and submitting proposals for new research projects that will address a myriad of issues related to prevention, screening, and treatment of the virus. In addition to new clinical trials of vaccine and drug treatments, our work will likely include research into how the virus affects physical and mental health, as well as how it impacts care for other conditions such as cancer, chronic pain, diabetes, and heart disease. We hope to conduct pharmaco-epidemiological studies of treatments that might work, studies of the social impacts of the pandemic, and more.
We plan to move as quickly as possible, using our unique research capabilities not only to attack COVID-19, but also to prepare our nation’s health care system for future pandemic threats.
The weeks ahead, with their inevitable losses, will not be easy. But I hope that we can take comfort in the work we’re doing together to weather this crisis and defeat the virus.
At a press briefing last week, NIAID Director Anthony Fauci, MD, called the vaccine trial we’re conducting at KPWHRI an “ultimate game changer” in this collective fight. I hope that in a year’s time we can look back at his words and see that he was right about this work and more.
Thanks to all of you for the work you’re doing. And thanks especially to the health care providers who are on the front lines of this battle.
Please take care and stay safe.
--Rita
co-researchers
Lisa A. Jackson, MD, MPH
Senior Investigator
,
research
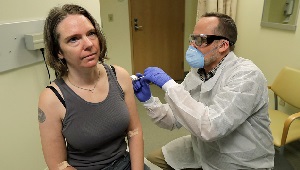
Kaiser Permanente launches first coronavirus vaccine trial
On March 16, 4 volunteers received an injection of an mRNA vaccine for COVID-19 in an NIH-funded trial in Seattle.
healthy findings

Ask an epidemiologist: Where to learn about COVID-19
There’s much confusion about the new disease, but numbers don’t lie. The challenge is finding the right ones.
Behavioral Health

Surviving an FFP: COVID-19
Dr. Jennifer McClure shares advice and resources for staying physically and emotionally well during the COVID-19 crisis, and beyond.


