Clinical trial will evaluate mpox vaccine for adolescents

The NIH-sponsored trial will help inform decisions about vaccine approval for 12- to 17-year-olds
In 2022, multiple countries, including the U.S., saw an outbreak of mpox, previously known as monkeypox. The mpox virus is related to smallpox but is much less deadly. However, it can cause serious symptoms, including fever, body aches, and skin lesions. The outbreak was declared a public health emergency in August 2022 and, to date, has affected more than 30,000 people in the U.S. and more than 85,000 globally.
Now, Kaiser Permanente Washington Health Research Institute (KPWHRI) is one of 18 sites participating in a clinical trial to evaluate the safety of a vaccine for mpox in adolescents as well as the immune response it generates. Sponsored by the National Institutes of Health (NIH), the trial will compare the immune response in people ages 12 through 17 with the response in those ages 18 through 50. The results will be used to help the U.S. Food and Drug Administration (FDA) make decisions about approving the vaccine for the younger age group.
The JYNNEOS vaccine is the only vaccine currently approved by the FDA for prevention of mpox, but it is approved only for adults. During the mpox outbreak, the FDA granted an emergency use authorization for providing the vaccine to adolescents 12 through 17 years of age who were at high risk of infection. Although rates of mpox in the U.S. have declined, recurring outbreaks are a possibility.
“It’s a public health priority to have a vaccine approved for use in those younger than 18,” said Lisa Jackson, MD, MPH, senior investigator at KPWHRI and principal investigator for the trial. “Although mpox has been rare in adolescents, there is the possibility that this age group could be at increased risk if we see another outbreak, so we want to make sure we have good data on how this vaccine works for them.”
The vaccine uses a weakened form of live virus that is not able to replicate and is also approved for prevention of smallpox. In 2010, the research institute was involved in a trial supporting intradermal use of the same vaccine (injecting the vaccine underneath the top skin layer).
Participating in the trial
Healthy adolescents ages 12 to 17 and adults ages 18 to 50 may be eligible to participate in this trial. Participation involves 2 injections of the JYNNEOS vaccine at the dose that is approved for adults, with up to 9 scheduled visits at the KPWHRI research clinic and 1 telephone visit over a period of 13 months. Vaccination visits will take about 3 hours, and additional clinic visits will last about 30 minutes. Participants will receive a $75 gift card for each clinic visit and free parking or transit tickets.
To learn more about this study in Seattle, email KPWA.vaccine@kp.org with your full name and date of birth and the best phone number and time to reach you.
Visit ClinicalTrials.gov for additional details. The study is sponsored by the National Institute of Allergy and Infectious Diseases (NIAID) at the NIH.
The study site is Kaiser Permanente Washington Health Research Institute — Vaccines and Infectious Diseases (UM1AI148576).
By Amelia Apfel
Research
New study confirms safety of shingles vaccine
KPWHRI researchers analyzed data from more than 640,000 vaccine doses to understand risk of severe reactions.
Research
Researchers begin clinical trial to assess schistosomiasis vaccine
Participant in KPWHRI trial has first-ever injection of SchistoShield vaccination.
1st vaccine shot
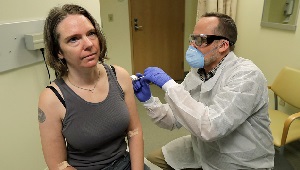
COVID-19 vaccine trial volunteer on the shot heard 'round the world
On March 16, 2020, Jennifer Haller became the first person to be injected with a COVID-19 vaccine in a clinical trial.



